Research Overview:
The apolipoprotein B mRNA editing enzyme catalytic polypeptide-like 3 (APOBEC3) proteins are a group of seven cytidine deaminases (A3A, A3B, A3C, A3D, A3F, A3G, A3H). APOBEC3-induced deamination results in the conversion of cytidines (C) to uridines (U). We discovered that the APOBEC3-induced mutagenesis is clonally enriched in chemotherapy-treated urothelial carcinoma and that its shape the tumor's evolution of throughout its lifetime (Faltas et al. Nature Genetics, 2016). We are currently using several model systems to dissect the role of APOBEC3-induced mutagenesis in promoting genomic-instability and driving tumorigenesis and chemotherapy-resistance in urothelial carcinoma.
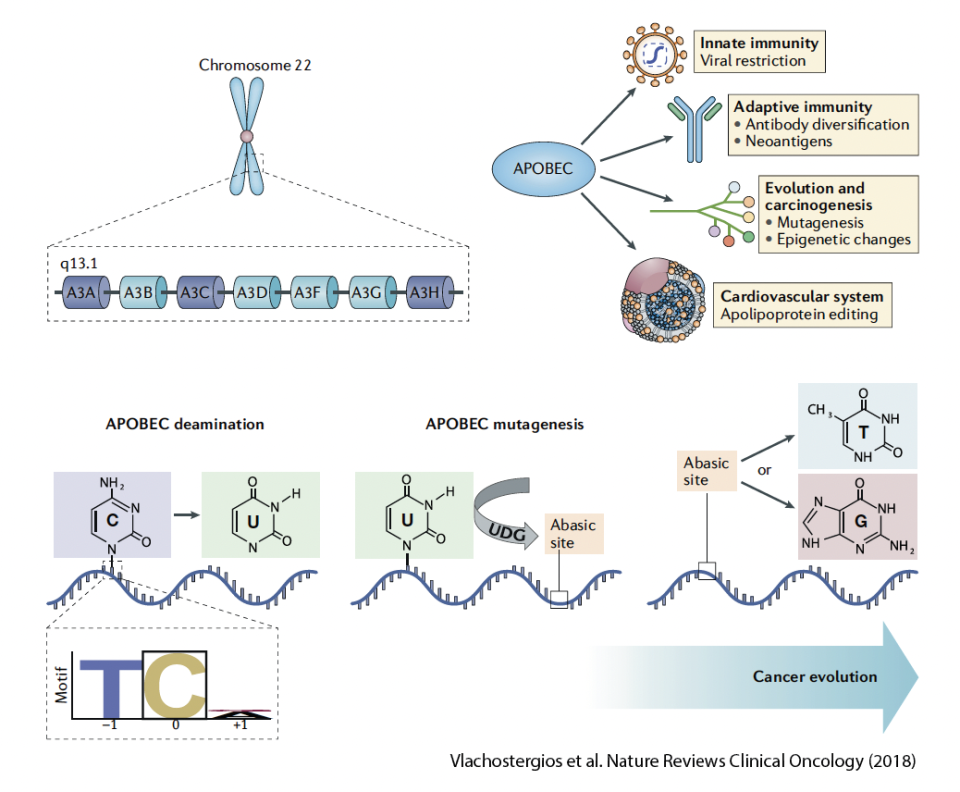
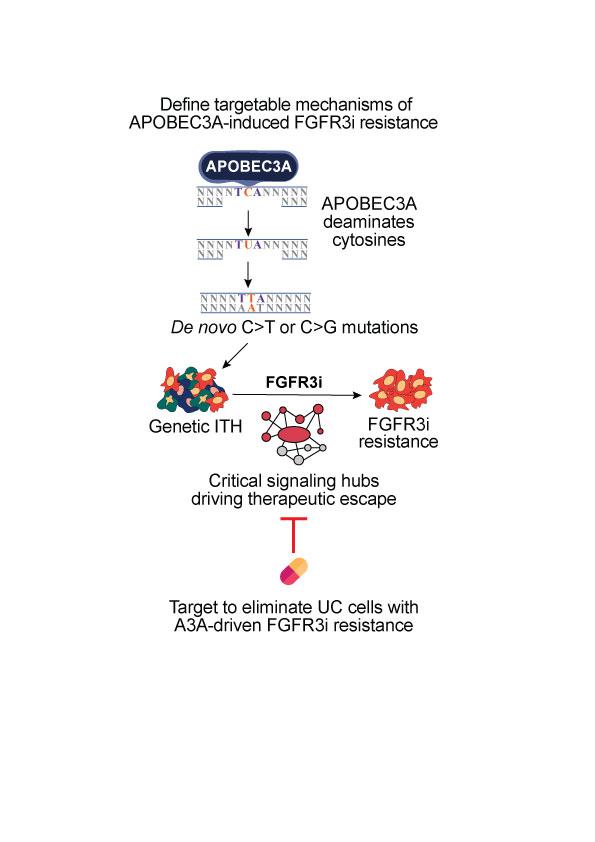
APOBEC3-induced drug-resistance
We are investigating the mechanisms by which APOBEC3 enzymes drive resistance chemotherapies and targeted therapies using a combination of cutting-edge experimental and computational methods.
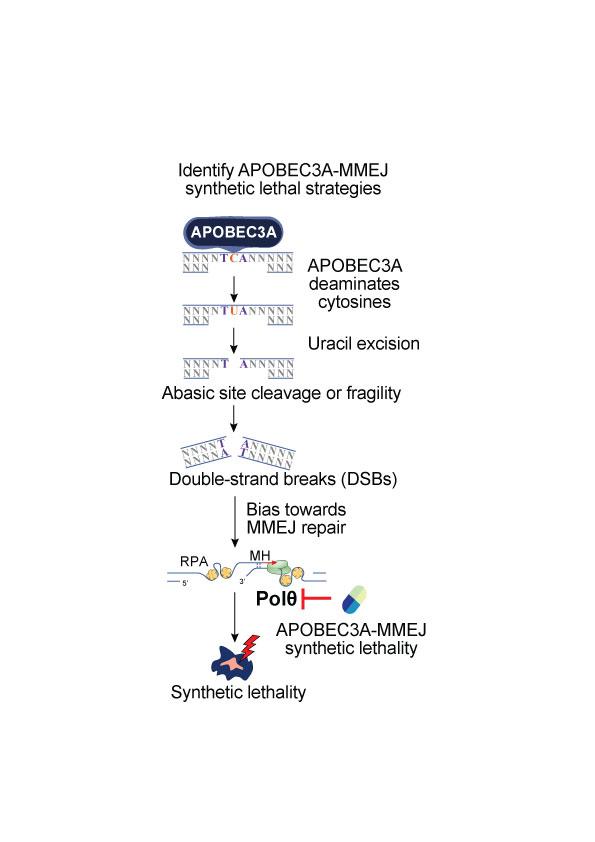
Identifying APOBEC3A-MMEJ synthetic lethal strategies
We are identifying synthetic lethal strategies to eliminate UC cells with A3A-induced DSBs using DNA damage repair reporter systems, biochemical methods, cell viability assays and animal models.
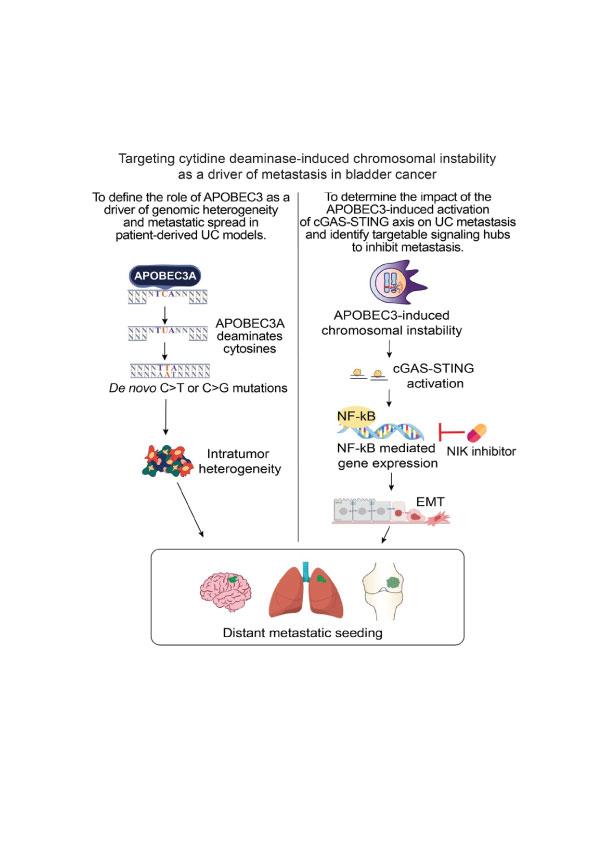
Targeting cytidine deaminase-induced chromosomal instability as a driver of metastasis in bladder cancer
We are determining the impact of the APOBEC3-induced activation of the cGAS-STING axis on UC metastasis and identify targetable signaling hubs to inhibit metastasis.
Bio-digital twins and AI-assisted clinical trials
We are investigating the use of multimodal deep-learning models integrating histopathology, cell type data, and molecular data to predict the outcomes of clinical trials. We are creating bio-digital twins that integrated multi-modal data to recapitulate and predict responses to cancer drugs in the clinic.
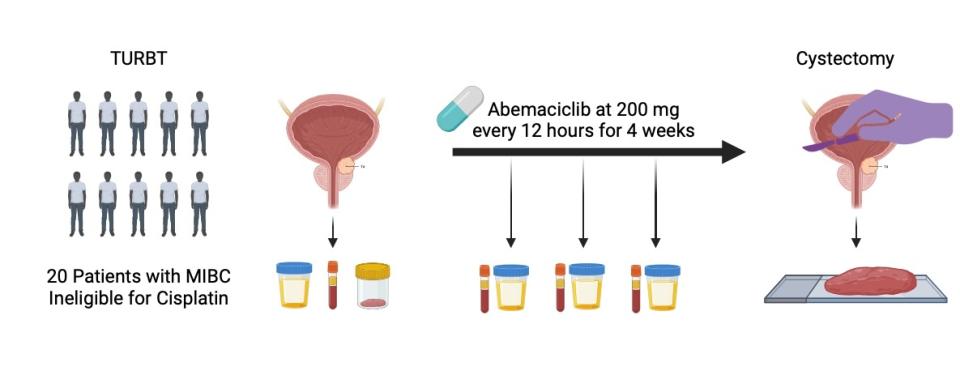
Cell cycLe inhibitiON to target the EVolution of urOthelial cancer (CLONEVO)
Dr. Faltas is leading the investigator-initiated Cell cycLe inhibitiON to target the EVolution of urOthelial cancer (CLONEVO), a single arm, window-of-opportunity trial of neoadjuvant abemaciclib which will evaluate the evolution of tumor cell and tumor microenvironment in response to CDK4/6 inhibition.
Meta-Track
The Meta-Track study will evaluate bladder cancer evolution using paired primary-metastatic organoids and humanized mouse models. This project aims to understand and modulate the unique biology underlying immune-tumor interactions permissive to metastatic spread in each patient.
UNO-BIO: Urothelium Normal Organoids Biobank
We are aiming to establish normal urothelial organoids from the urine of patients with high-risk germline mutations or unique environmental exposures that predispose them to bladder cancer.